Bicarbonate, also known as hydrogen carbonate, is a chemical compound that plays a vital role in maintaining the acid-base balance in the human body. It is an anion (negatively charged ion) formed when carbon dioxide dissolves in water. Bicarbonate is present in various bodily fluids, including blood, saliva, and urine, and it performs several essential functions to ensure proper physiological functioning. In this article, we will explore the role of bicarbonate in the body, focusing on its importance and the systems it affects.
Acid-Base Balance
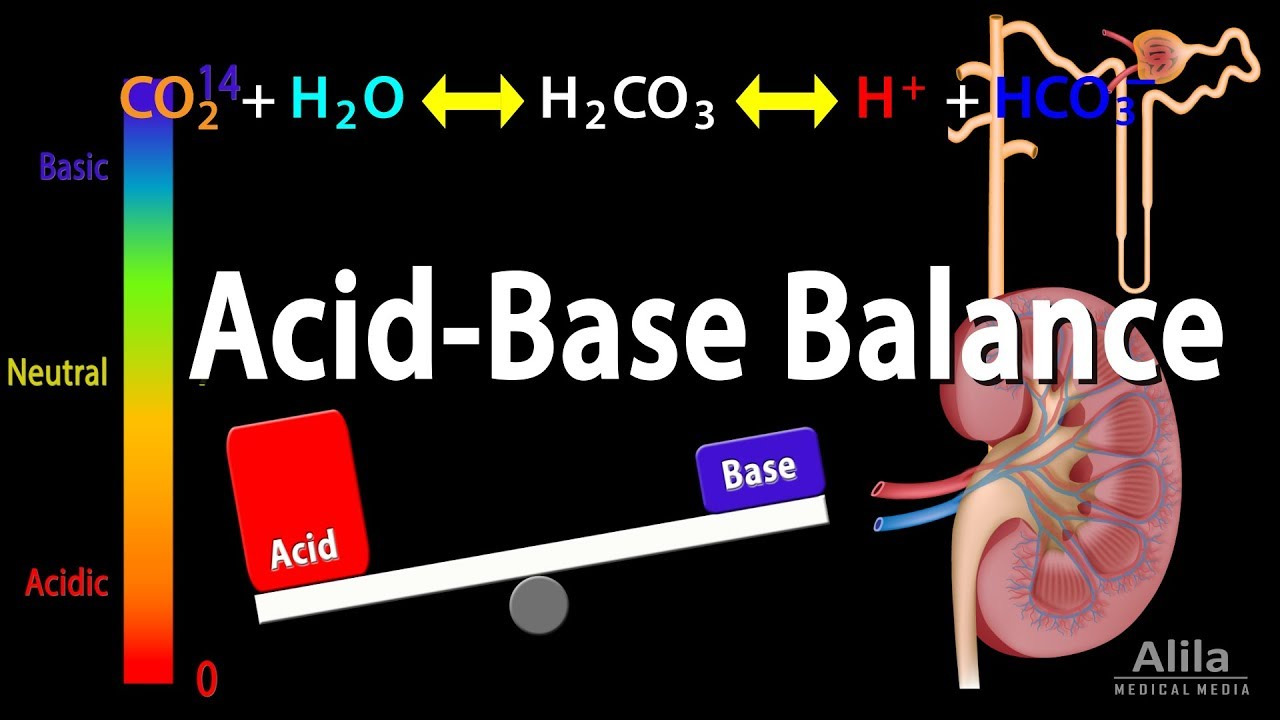
One of the primary functions of bicarbonate in the body is to regulate the acid-base balance (remember, if you buy bikarbonat storpack, you’ll save more money.) The body strives to maintain a specific pH level, which is a measure of acidity or alkalinity. Bicarbonate acts as a buffer, meaning it helps maintain the pH within a narrow range by neutralizing excess acid or base. It accomplishes this by accepting or releasing hydrogen ions (H+), depending on the body’s needs. This buffering action is crucial for various physiological processes to occur optimally.
Respiration and Carbon Dioxide Transport
Bicarbonate is intricately involved in the process of respiration and the transport of carbon dioxide (CO2) in the body. When we breathe, oxygen is taken in, and carbon dioxide is released. Carbon dioxide produced in tissues diffuses into the bloodstream and combines with water to form carbonic acid (H2CO3). Carbonic acid quickly dissociates into bicarbonate and hydrogen ions. Bicarbonate is then transported in the plasma to the lungs, where it is converted back into carbon dioxide for exhalation. This mechanism allows for the efficient removal of carbon dioxide waste from the body and helps maintain the acid-base balance.
Digestion and Enzymatic Function
Bicarbonate is also involved in the digestive process. It is released by the pancreas into the small intestine to neutralize stomach acid (hydrochloric acid) and create a suitable pH environment for the activity of digestive enzymes. This neutralization is crucial for optimal enzyme function, allowing for the breakdown and absorption of nutrients from food. Bicarbonate ensures that the acidic chyme from the stomach is adequately neutralized before it enters the small intestine.
Kidney Function and Acid Excretion
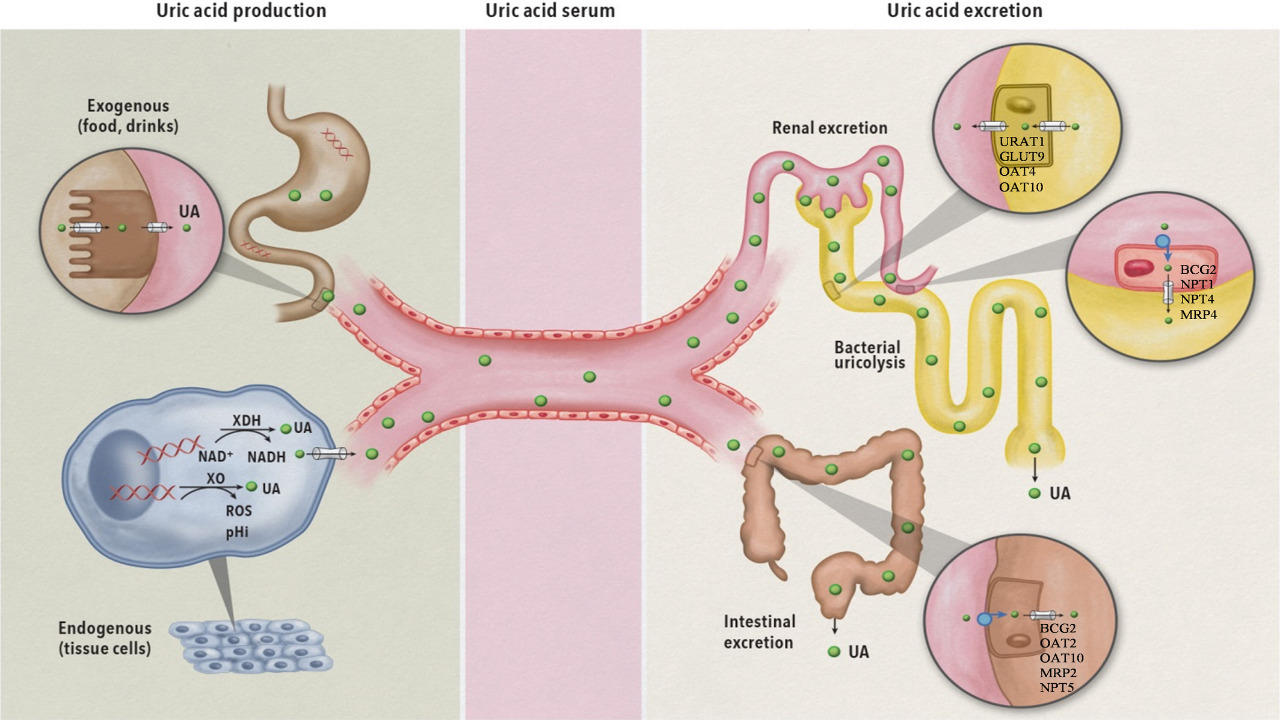
The kidneys play a vital role in maintaining acid-base balance by regulating bicarbonate levels in the body. They selectively reabsorb bicarbonate from the urine and release it into the bloodstream, helping to replenish bicarbonate levels when needed. Additionally, the kidneys excrete excess acid (hydrogen ions) in the urine, which helps maintain a proper pH balance. This renal regulation of bicarbonate and acid excretion is essential for the overall acid-base homeostasis of the body.
Electrolyte Balance and Nerve Function
Bicarbonate is an important electrolyte that contributes to the overall balance of ions in the body. It works in conjunction with other electrolytes, such as sodium, potassium, and chloride, to maintain proper nerve function and cellular communication. Bicarbonate ions are involved in nerve impulse transmission and help regulate the electrical activity of muscles, including the heart. Imbalances in bicarbonate levels can disrupt these processes and lead to various health issues.
Conclusion
Bicarbonate plays a crucial role in maintaining the acid-base balance in the human body. Its ability to act as a buffer, transport carbon dioxide, support digestion, regulate kidney function, and contribute to electrolyte balance and nerve function highlights its significance. Any disruption in bicarbonate levels can have profound effects on physiological processes. Understanding the role of bicarbonate underscores the importance of maintaining a healthy acid-base balance for overall well-being.
